 
Those four elements are numbered 113, 115, 117 and 118. That means the seventh period of the periodic table is complete. In 2016, four new elements were added to the periodic table. When was the periodic table of elements last updated? The elements on the right-hand side are generally non-metals. For example, elements on the left-hand side of the periodic table are generally metals. The table can also give you information on chemical properties. This design lets you quickly find an element’s symbol, atomic number, and atomic mass. The periodic table that we use today contains seven periods and 18 groups. Here is an image to help you remember that groups run vertically and periods run horizontally (© 2019 Let’s Talk Science). The rows of the periodic table are called periods. They are arranged based on their atomic number, electron configurations, and chemical properties. The periodic table of elements is a table that arranges the chemical elements in a logical way. They orbit around the nucleus.Ītoms of a certain element have the same number of protons but can have different numbers of neutrons. But like protons, they are found in the nucleus.Įlectrons have a negative electrical charge. Unlike protons, neutrons do not have any electrical charge. Neutrons are a type of particle with approximately the same mass as a proton. Various ways to describe a carbon atom, including a Lewis structure, periodic table entry and Bohr model (Sources: Let’s Talk Science based on image from K!roman, SVG: Marlus_Gancher via Wikimedia Commons, me via Wikimedia Commons, modification of Ahazard.sciencewriter via Wikimedia Commons).Įlements can also contain neutrons and electrons. The nucleus is a dense region located in the center of an atom. Protons have a positive electrical charge and are contained in the nucleus. For example, carbon has an atomic number of 6. The atomic number tells you the number of protons in an element. You can find it in water (H 2O) and many other molecules that living organisms rely on.Įach element has its own atomic number. It is the third-most abundant element in the universe. They make up all of the ordinary matter of the universe. What are chemical elements?Ĭhemical elements are the building blocks of chemistry. But first, let’s learn about what chemical elements are. Let’s learn a little bit about these newest elements, and what they were named. In 2016, four new elements were added to the periodic table of elements. However, once a name and symbol have been chosen, they can never be changed. They say that elements can be named after mythical characters, concepts, minerals, places, element properties, or scientists. NAME OF ELEMENT WITH ATOMIC NUMBER 5 TVIf you discovered a new element, what would you name it? Would you name it after your favourite TV character? Your hometown? Yourself? The International Union of Pure and Applied Chemistry (IUPAC) sets guidelines on naming chemical elements. NAME OF ELEMENT WITH ATOMIC NUMBER 5 PROFESSIONALOpen Professional Learning × Close Professional Learning Such an arrangement helps explain the periodicity and periodic trends observed across the elements of the periodic table.Open Educational Resources × Close Educational Resources The N shell containing 4s, 4d, 4p and 4f, can carry 32 electrons. The M shell contains 3s, 3p, and 3d, and can carry 18 electrons. The K shell contains a 1s subshell hence it can carry 2 electrons, the L shell has 2s and 2p, and can carry 8 electrons. This decides the electron capacity of the shells. The maximum electrons that can be carried by the sub-shell S is 2, by P is 6, by D is 10, and the F sub-shell can carry 14. Each shell and subshell have a limitation on the amount of electrons that it can carry. The subshells have a distinct shape and configuration, in which the electrons move freely. They stand for sharp (S), principal (P), diffuse (D), and fundamental (F). The shells are labeled K, L, M, N, and so on, from the innermost to the outermost shell.Įach shell has subshells that are named for the type of emission lines produced from different states of angular momentum. This model has been widely accepted, and according to it, each atom has shells, which further have subshells. It involves the specific arrangement of electrons in shells and sub-shells of Bohr’s atomic model. 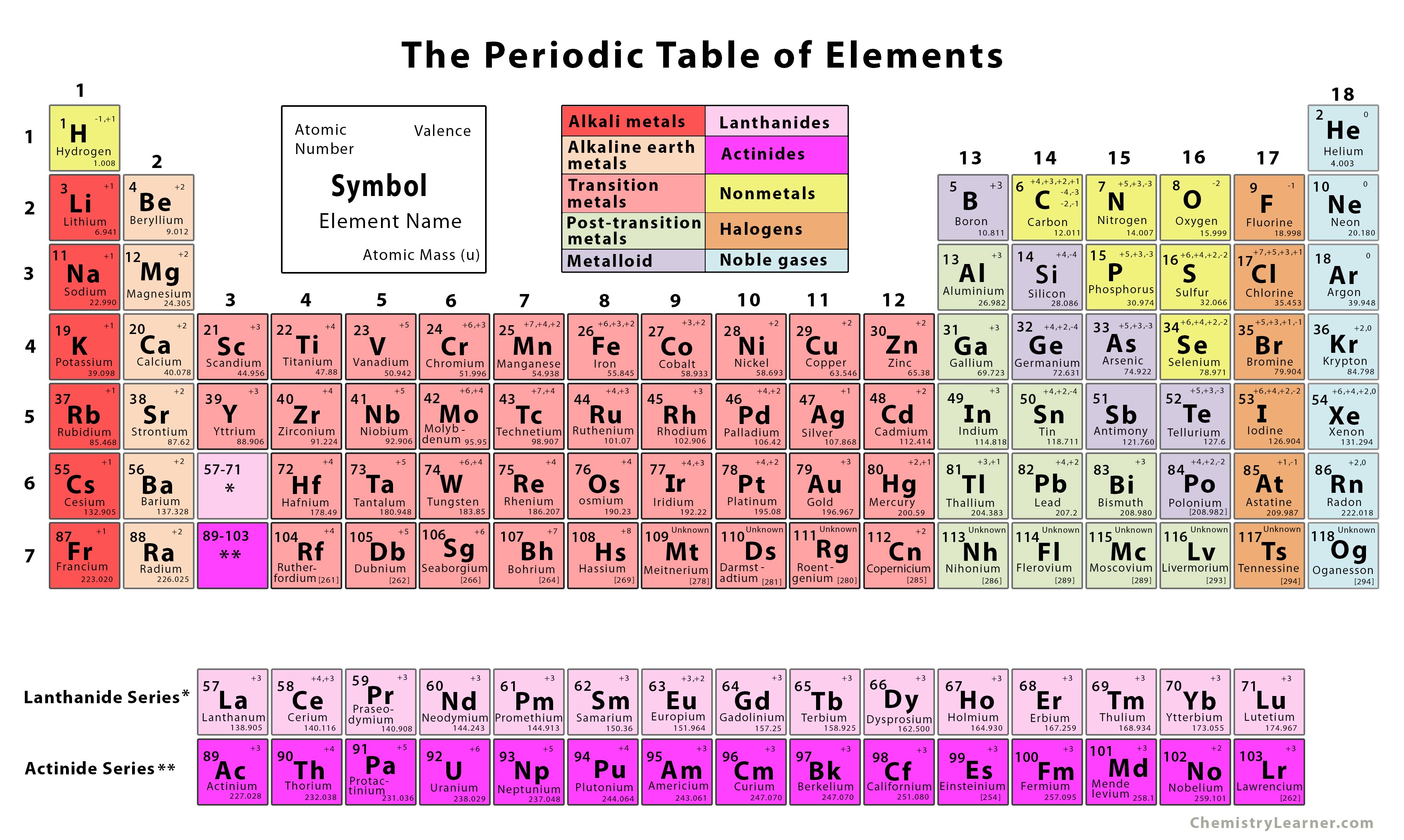
The concept of electronic configuration has replaced the older concept of valency and valence electrons. The electronic configuration of each element is decided by the Aufbau principle which states that the electrons fill orbitals in order of increasing energy levels. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
